Homeopathy & The jab
general & local improvements post cranial nerve damage - a peer reviewed case study
Influenza A associated Binocular Diplopia and concomitant accelerated myopia post COVID-19 mRNA Biological Injection.1
A case report by Sarah Penrose.
Published in Similia the Australian Journal of Homeopathic Medicine 36:2,Dec 2023.
ABSTRACT
COVID-19 mRNA Biological Injections have been shown to be detrimental to human health across a wide range of clinical domains, with side effects typified by immune mediated inflammation. This case outlines reversal of sudden and debilitating influenza A associated binocular diplopia and deteriorating myopia in a COVID-19 mRNA Biological Injection recipient during treatment with classical individualized homoeopathy in accordance with Direction of Cure theory. Controlled studies are required to establish the extent to which homoeopathy may be considered a potential therapeutic modality where pharmaceutical interventions manifest sequelae and iatrogenic disease.
Keywords
Binocular diplopia – COVID-19 mRNA Biological Injection – Homeopathic therapy - Case report.
INTRODUCTION
Binocular diplopia (double vision) is a misalignment of vision commonly due to a disorder of cranial nerves III, IV, and VI innervating the extraocular muscles, and may be isolated, idiopathic, or resulting from cranial nerve nuclei or infranuclear nerve disorders.2 Influenza A induced oculomotor nerve palsy is rare in adults and sparsely documented in medical literature.3
The COVID-19 mRNA Biological Injection has the potential to induce neurologic sequelae4 and oculomotor palsies,56 including binocular diplopia.7 The pathophysiology of post-immunization ocular nerve palsies and cranial neuropathies is unknown but hypothesized as relating to immune-mediated demyelination or localized vasculitis8 with typical onset ranging from days to weeks post-immunization.9
Medical literature has documented homoeopathy resulting in good outcome with no residual clinical signs of the disease Myasthenia gravis10 which is characterized by ocular symptoms leading to diplopia.11
Case report - Thirty-three-year-old West Australian woman.
Initial consultation - 18th October 2022.
Main complaint Sudden binocular diplopia onset during influenza A infection in April 2022, with deteriorating vision beginning four weeks’ post administration of a BioNTech/Pfizer COVID-19 mRNA Biological Injection (C19MBI) taken February 2022.
Background Myopic since age 10, stronger lenses were prescribed during a September 2021 eye examination which required replacement after just six months, in March 2022, when cranial nerve VI damage was diagnosed by an ophthalmologist who advised increasingly stronger prescription lenses as a likelihood, and subsequently diagnosed binocular diplopia in May 2022. Additional symptoms of chest pain and palpitations (inconclusive electrocardiogram results), right shoulder bursitis with shooting pain down arms and chest, frontal and temple headaches, and alteration in menses developed within days of receiving C19MBI and had all resolved without treatment by March 2022.
Table 1: Presenting symptoms October 2022
Figure 1: Repertorization October 2022
Figure 2: Repertorization results October 2022
Gelsemium sempervirens Vithoulkas states that immobilizing anticipatory fear is central to a Gelsemium case. The characteristic paralysis and weakness make Gelsemium one of our main remedies for neurological damage. Complaints after influenza. Double vision. Paralysis of optic nerve. Things seem double on looking sideways. Diplopia when inclining the head to either side. Concentration difficult.12
The first documented homeopathic pathogenetic trial of Gelsemium was conducted ‘on at least fifty people’ by Dr. Douglass of Milwaukee13 and published in Dr. Hale’s Monograph in 1862. Dr. Edgar Holden’s published sphygmograph experiments with potentized Gelsemium were read before the Medical Society of New Jersey14 - the sphygmograph was one of the first precision cardiology instruments.15
Prescription October 2022 Gelsemium 30c 1x daily.
Posology Degenerative chronic disease and absence of fever in response to an infective agent (influenza A in this case) is due to a compromised immune system and indicates a Group C Level of Health classification where a 30c potency should be repeated every day unless / until there is an aggravation. Level seven may manifest acute diseases which are mild and unaccompanied by high fever (over 38.5°C).16
Reaction to the remedy October 2022 ‘My vision went a bit funny in my right eye for a couple of days and I had a really, really flat day, no energy, terrible mood, didn’t want to see or speak to anyone. The ‘I can’t be bothered’ mentality. I don’t feel any different and I didn’t think there was change in my double vision but I’ve just done a little test of looking forward and moving my head to each side and I can actually turn my head a lot further whereas even the slightest turn would set off double vision.’
Gelsemium 30C was discontinued after three doses. An initial aggravation followed by amelioration of the local complaint suggests a functioning defence system where no remedy(s) should be prescribed for minor symptomatology alongside a generally improving state.17
Table 2: Follow-up Appointments and Prescriptions Nov. 2022 – Sept. 2023

Table 3: Ameliorated, Residual and New Symptoms as of September 2023
Discussion In this case, the initial aggravation of symptoms followed by general and local improvements suggest a well-functioning defence system. As outlined (Table 2), repetition of the remedy in an effort for speedy resolution is not advised18 as the defence system (in this case) is able to cope with symptoms which only once they became extremely bothersome were cautiously prescribed upon and the remedy repeated. Amelioration of the presenting symptoms accompanied by the return of old, suppressed symptoms (Table 3) as the general chronic condition improves is indicative of deep and positive change occurring within the defence system in accordance with Direction of Cure theory.19
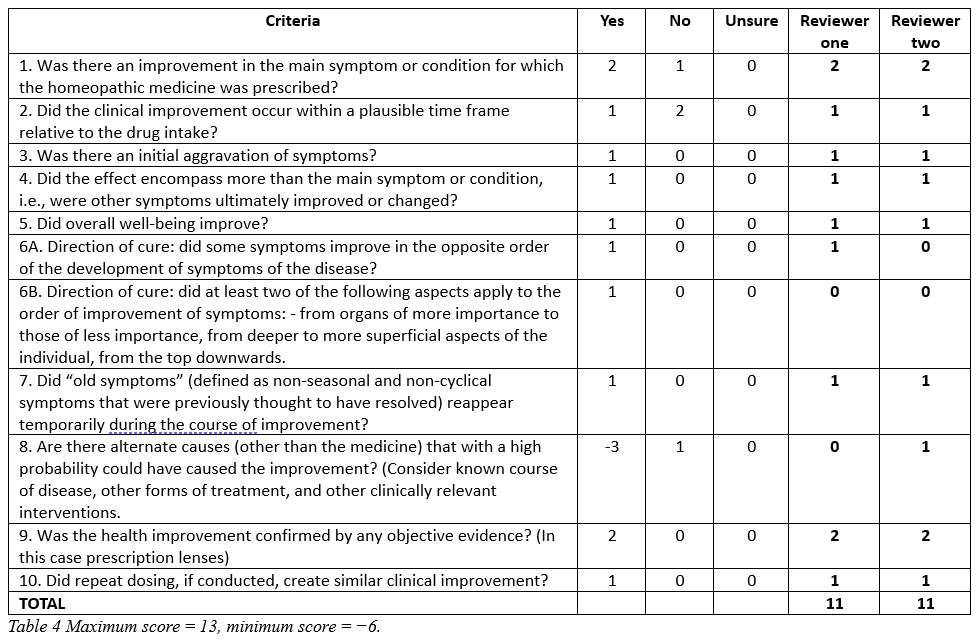
Case reports are hierarchically weak evidence of causality not suited to investigate therapeutic efficacy however, the Modified Naranjo Criteria for Homoeopathy may assess how likely causal attribution is to the final clinical outcome.20
Table 4: Modified Naranjo Criteria for Homoeopathy – for causality assessment.
The COVID-19 mRNA Biological Injection Decreased bacterial and viral stimulant -induced cytokine responses are linked to C19MBI administration,21 as are neuro-ophthalmic complications,22 and transient local and systemic reactions, and tissue and organ damage and failure, likely induced by Liquid Nano Particles causing elaborate immune responses23 - the cationic/ionizable lipid component of which is both inflammatory and cytotoxic.24 C19MBI are highly volatile compounds capable of inducing serious burns from metres away25 fueling speculation that it is this volatility driving the shedding / spreading mechanism.
Incidence of COVID-19 infection increases with each subsequent C19MBI,262728 and as for example., C19MBI recipients have a thirteen-fold increased risk of developing a COVID-19 infection when compared to individuals who acquired natural immunity through prior infection,29 a doubled risk of retinal vascular occlusion,30 and a one in thirty five chance post C19MBI booster of vaccine-associated myocardial injury,31 it has been suggested that worldwide regulatory agency capture continues to drive an ongoing campaign of medical misinformation.32
Authors note This case has been checked for accuracy and given consent to publish. Gratitude is expressed to MONARCH reviewers Dr. Seema Mahesh BHMS, MD(hom), Dip IACH, MSc (Medical sciences) for salient editorial insights and encouragement, and Barbara Roberts RCHom, BPharm (hons), DipHom.
Penrose, S. 2023. Influenza A associated Binocular Diplopia and concomitant accelerated myopia post COVID-19 mRNA Biological Injection. Similia, The Australian Journal of Homeopathic Medicine, 36:2. Hobart. The Australian Homeopathic Association.
Merck et al., Diplopia (double vision). Merck Manual Professional Version. [Internet]. 2023 [cited 2023 Feb 08] Available from https://www.merckmanuals.com/professional/eye-disorders/symptoms-of-ophthalmologic-disorders/diplopia
Senda et al., Acute Unilateral Isolated Oculomotor Nerve Palsy in an Adult Patient with Influenza A. Intern Med. [Internet]. 2019 [cited 2023 Jul 24]. Feb 1;58(3):433-436. Available from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6395116/
Wan et al. Bell's palsy following vaccination with mRNA (BNT162b2) and inactivated (CoronaVac) SARS-CoV-2 vaccines: a case series and nested case-control study. Lancet Infect Dis. [Internet]. 2021 [cited 2023 Feb 08]; Jan;22(1):64-72. Available from https://pubmed.ncbi.nlm.nih.gov/34411532/
Bolletta et al., Uveitis and Other Ocular Complications Following COVID-19 Vaccination. J Clin Med. [Internet]. 2021. [cited 2023 Jul 24]. Available from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8704915/
Ng et al., Ocular Adverse Events After COVID-19 Vaccination. Ocul Immunol Inflamm. [Internet]. 2021 [cited 2023 Jul 24]. Available from https://pubmed.ncbi.nlm.nih.gov/34559576/
Mahgerefteh et al., Binocular Horizontal Diplopia Following mRNA-1273 Vaccine. J Neuroophthalmol. [Internet]. 2022 [cited 2023 Feb 08]; 2022 Apr 27. Available from https://pubmed.ncbi.nlm.nih.gov/35482435/
Woo et al., Motor palsies of cranial nerves (excluding VII) after vaccination: reports to the US Vaccine Adverse Event Reporting System. Hum Vaccin Immunother. [Internet]. 2014 [cited 2023 Feb 08];10(2):301-5. Available from https://pubmed.ncbi.nlm.nih.gov/24231288/
Bolletta et al., Uveitis and Other Ocular Complications Following COVID-19 Vaccination. J Clin Med. [Internet]. 2021. [cited 2023 Jul 24]. Available from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8704915/
Văcăraș et al., Myasthenia gravis therapy with individualized homoeopathy: A case report. Clinical Case Reports. [Internet]. 2020 [cited 2023 Feb 08]; Jul 29;8(12):2464-2468. Available from https://onlinelibrary.wiley.com/doi/10.1002/ccr3.3190
Smith & Lee. Update on Ocular Myasthenia Gravis. Neurol Clin. [Internet]. 2017 [cited 2023 Feb 08];Feb;35(1):115-123. Available from https://pubmed.ncbi.nlm.nih.gov/27886889/
Vithoulkas, G. Materia Medica Viva Volume 11 Ferrum Metallicum to Helleborous Niger. Alonissos: The international Academy of Classical Homoeopathy; 2012 2nd ed. (Originally published 2006).
Allen, T. The Encyclopaedia of Pure Materia Medica. A record of the positive effects of drugs upon the healthy human organism. Vol. IV. 6th Ed. New Delhi: B. Jain Publishers; 2011. (Originally published in 1874)
Miccant. Materia Medica. Hale; Therapeutics of New Remedies. ISIS [Computer program]. Great Britain. 2015.
Moss. The sphygmograph in America: writing the pulse. Am J Cardiol. [Internet]. 2006 [cited 2023 Aug 05];Feb 15;97(4):580-7. Available from https://pubmed.ncbi.nlm.nih.gov/16461061/
Vithoulkas, G. Levels of Health, The second volume of the Science of Homoeopathy, revised edition. Alonissos: International Academy of Classical Homoeopathy; 2017.
Vithoulkas, G. Levels of Health, The second volume of the Science of Homoeopathy, revised edition. Alonissos: International Academy of Classical Homoeopathy; 2017.
Vithoulkas, G. Levels of Health, The second volume of the Science of Homoeopathy, revised edition. Alonissos: International Academy of Classical Homoeopathy; 2017.
Mahesh et al., Individualized homeopathic therapy in ANCA negative rapidly progressive necrotising crescentic glomerulonephritis with severe renal insufficiency – a case repot. J M Life. [Internet]. 2019. [cited 2023 Feb 08] Jan-Mar;12(1):49-55. Available from https://pubmed.ncbi.nlm.nih.gov/31123525/
Lamba et al. Evaluation of the Modified Naranjo Criteria for Assessing Causal Attribution of Clinical Outcome to Homeopathic Intervention as Presented in Case Reports. Homoeopathy. [Internet]. 2020. [cited 2023 May 08] Nov;109(4):191-197. Available from: https://www.thieme-connect.com/products/ejournals/html/10.1055/s-0040-1701251#JR1900046-10
Noe et al., BNT162b2 vaccination is associated with a decrease in bacterial and viral stimulant -induced cytokine responses one month after vaccination. Front. Immunol. [Internet]. 2023. [cited 2023 Aug 31]. 14:1242380. https://www.frontiersin.org/articles/10.3389/fimmu.2023.1242380/full
Chaitanuwong et al., SARS-CoV-2 Infection, Vaccination, and Neuro-Ophthalmic Complications. J Neuroophthalmol. [Internet]. 2023. [cited 2023 Sept 16] Mar 1;43(1):1-4. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9924739/
Ndeupen et al., The mRNA-LNP platform's lipid nanoparticle component used in preclinical vaccine studies is highly inflammatory. iScience. [Internet]. 2021. [cited 2023 Feb 08] Dec 17;24(12):103479. Available from https://www.cell.com/iscience/fulltext/S2589-0042(21)01450-4?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS2589004221014504%3Fshowall%3Dtrue
Samaridou et al., Lipid nanoparticles for nucleic acid delivery: Current perspectives. Adv. Drug Deliv. Rev. [Internet]. 2020. [cited 2023 Feb 08] 154–155, 37–63. Available from: https://pubmed.ncbi.nlm.nih.gov/32526452/
Chantra et al., Ocular Surface Erosion after Suspected Exposure to Evaporated COVID-19 Vaccine. Case Rep Ophthalmol. [Internet]. 2021. [cited 2023 Feb 08] Dec 2;12(3):944-951. Available from https://www.karger.com/Article/FullText/520500
Shrestha et al. Effectiveness of the Coronavirus Disease 2019 (COVID-19) Bivalent Vaccine. medRxiv. [Internet]. 2022 [cited 2023 Feb 08]; https://www.medrxiv.org/content/10.1101/2022.12.17.22283625v1
Boucau et al., Duration of Shedding of Culturable Virus in SARS-CoV-2 Omicron (BA.1) Infection. N Engl J Med. [Internet]. 2022 [cited 2023 Feb 25]; Jul 21;387(3):275-277. Available from https://www.nejm.org/doi/full/10.1056/nejmc2202092
Marking et al., Correlates of protection, viral load trajectories and symptoms in BA.1, BA.1.1 and BA.2 breakthrough infections in triple vaccinated healthcare workers. MedRxiv. [Internet]. 2022 [cited 2023 Feb 25]; Preprint. Available from: https://www.medrxiv.org/content/10.1101/2022.04.02.22273333v2
Gazit et al., Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Naturally Acquired Immunity versus Vaccine-induced Immunity, Reinfections versus Breakthrough Infections: A Retrospective Cohort Study. Clinical Infectious Diseases [Internet]. 2022. [cited 2023 Sep 04]. Vol 75, 1,1 July, e545–e551. Available from: https://academic.oup.com/cid/article/75/1/e545/6563799?login=false
Li et al., Risk assessment of retinal vascular occlusion after COVID-19 vaccination. NPJ Vaccines. [Internet]. 2023. [cited 2023 Sep 04]. May 2;8(1):64. Available from: https://pubmed.ncbi.nlm.nih.gov/37130882/
Buergin et al., Sex-specific differences in myocardial injury incidence after COVID-19 mRNA-1273 booster vaccination. Eur J Heart Fail. [Internet]. 2023. [cited 2023 Sep 04]. Jul 20. Epub ahead of print. https://pubmed.ncbi.nlm.nih.gov/37470105/
Malhotra, A. Curing the pandemic of misinformation on COVID-19 mRNA vaccines through real evidence-based medicine - Part 2. Journal of Insulin Resistance. [Internet]. 2022 [cited 2023 Feb 08]; 5(1). Available from https://insulinresistance.org/index.php/jir/article/view/72/228